Services
Independent, integrated respiratory product development. From ab initio formulation to Phase 1 clinical imaging, all performed on-site at our Cardiff Medicentre facility.
Fast2Clinic™
Rapid small-scale clinical manufacture and next-day release designed to accelerate Phase 1 first-in-human studies without compromising GMP standards.
Nasal & Inhalation Formulation
From ab initio development to proof of concept, tailoring each formulation to your specific molecule, target patient profile, and chosen delivery format.
Analytical Excellence
Robust data generation through industry-standard cascade impactor testing, HPLC, spectroscopy, and rigorous impurity profiling.
Gamma Scintigraphy
In-house imaging suite integrated within a 48-bed CRO, delivering quantitative in vivo deposition data to validate respiratory delivery platforms.

Fast2Clinic™
i2c stands out for its unique ability to deliver both laboratory and pilot-scale batches. Our flexible, scalable approach ensures highly efficient transitions from lab to clinic, backed by agile, client-focused project management.

Nasal & Inhalation Formulation
From ab initio development to proof of concept, i2c supports every aspect of inhalation product development, spanning pMDIs, dry powder formulations, nebulisers, and nasal sprays. We tailor each formulation precisely to your molecule, chosen delivery format, and target patient profile.

Analytical Excellence
At i2c Pharmaceutical Services, analytical science is at the heart of everything we do. Our team provides the rigorous data and insight you need to accelerate your inhalation product development.
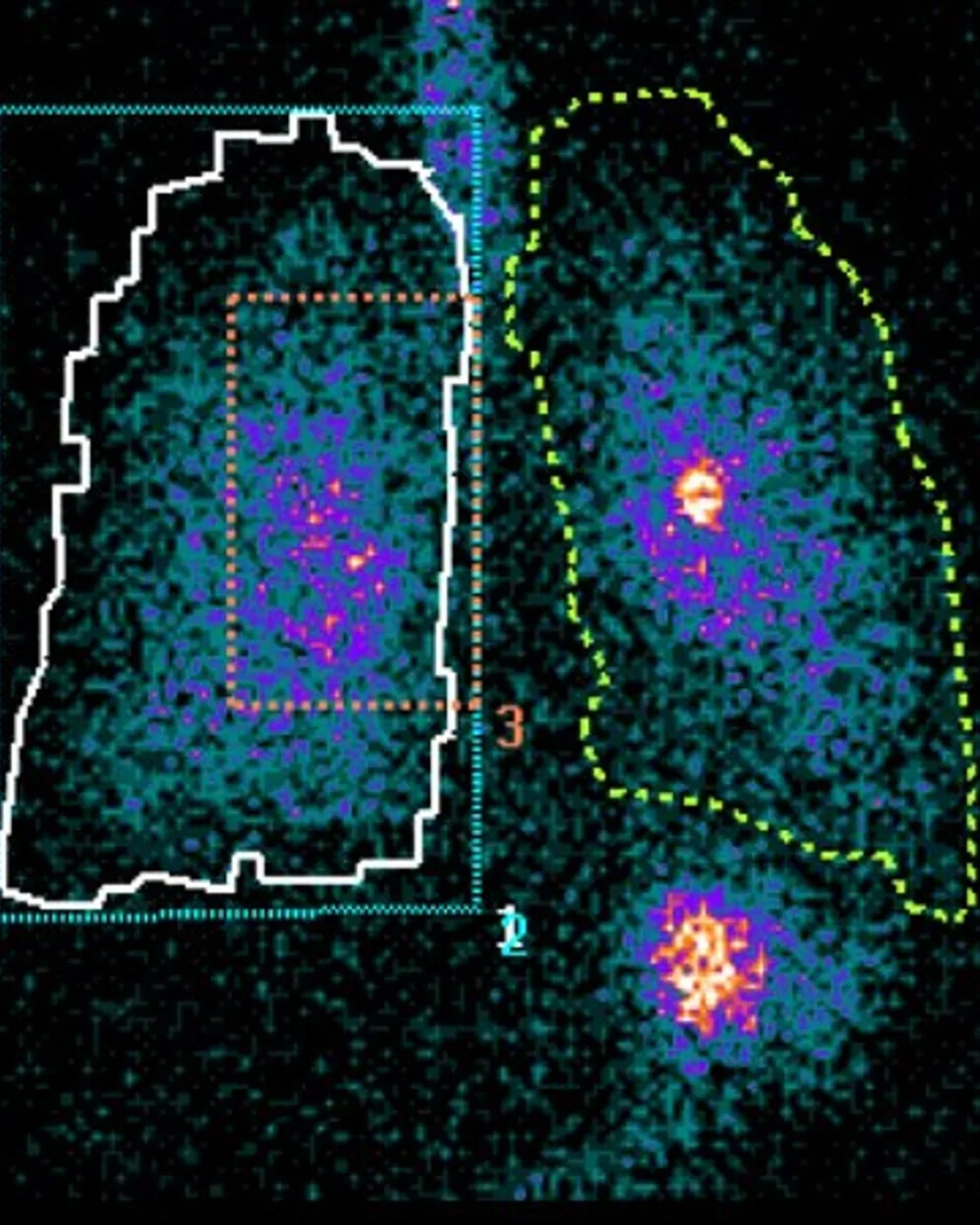
Gamma Scintigraphy
Gamma scintigraphy is a non-invasive imaging technique that uses radiolabelled drug formulations to visualize and quantitatively assess how much of an inhaled medicine is deposited in the respiratory tract. It enables direct measurement of lung, oropharyngeal, and gastrointestinal deposition, as well as clearance and dispersion over time.
Discover Respitab®
A revolutionary solid-state tablet platform for pressurized metered dose inhalers. By eliminating complex suspension mixing, Respitab® streamlines production, enhances batch uniformity, and significantly accelerates your route to clinic.
Visit Respitab.co.ukAn Integrated Approach
By unifying analytical testing, formulation, and clinical imaging within our state-of-the-art Cardiff facility, we eliminate the friction of multi-vendor management and completely accelerate your route to the clinic.
Formulation & Delivery
Expertise spanning pMDIs, dry powder formulations, nebulisers, and nasal sprays. We tailor every formulation precisely to the molecular requirements of your target patient profile.
Analytical Rigour
State-of-the-art instrumentation delivering robust aerosol characterisation, impurity profiling, and method development to support swift, compliant regulatory progression.
In Vivo Validation
Gold-standard gamma scintigraphy imaging provides direct, highly quantitative data on pulmonary deposition, offering critical validation prior to late-stage clinical trials.
