From Innovation to Clinic Bespoke R&D Pathways
i2c Pharm is an independent inhalation CRO providing formulation development, analytical services, gamma scintigraphy, and patented Respitab® MDI technology supporting global pharma from first formulation to clinical proof with our Fast2Clinic approach.
About i2c
i2c is an independent CRO based in Cardiff, Wales specialising in inhalation formulation development. From early feasibility through to Phase I clinical supply, we support global pharma and medtech clients under one integrated roof.
inhalation R&D
served
& On-site CRO

Our Services
View all services →
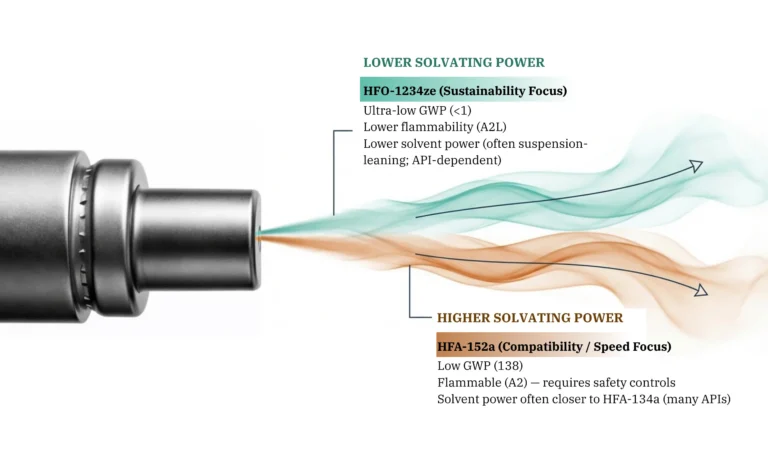
MDI Technology
Our patented tablet-based platform for pMDI development. Eliminates high-shear mixing, improves batch uniformity, and accelerates your MDI programme.
Discover Respitab™ →
Rapid small-scale manufacture of just-in-time clinical formulations. Cut timelines and get your product to Phase I faster.
Learn More →
Inhalation Formulation
Expertise across pMDIs, DPIs, nebulisers, and nasal sprays. Each formulation tailored to your molecule and target patient profile.
Learn More →
Excellence
Full-spectrum testing using NGI, Andersen, HPLC, and DUSA systems. Reliable, reproducible data to support every formulation decision.
Learn More →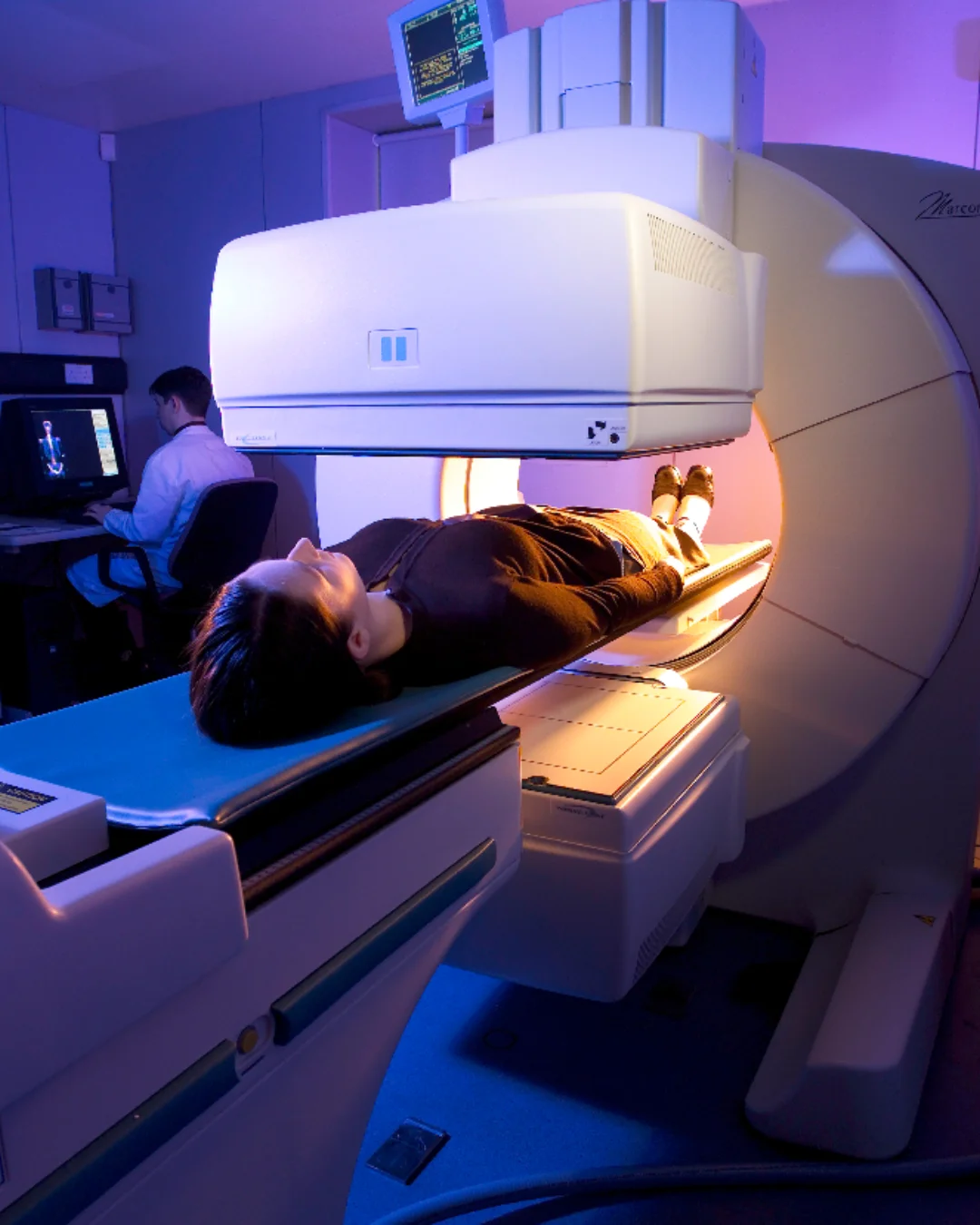
Scintigraphy
Gold-standard non-invasive imaging to quantify inhaled drug deposition in the lungs, oropharynx, and GI tract. Trusted data for regulatory submissions.
Learn More →Why Choose i2c
Independent, integrated, and built entirely around inhalation.

Expertise
30+ years focused exclusively on inhaled drug development.

Technology
Patented Respitab® MDI platform

Offering
Scintigraphy, formulation & manufacture under one roof.

Depth
NGI, HPLC, DUSA & Andersen, regulatory-grade data every time.

Focused
Direct access to scientists, no account manager layer.
Research Foundation & hypothesis
Testing pMDI, DPI, Nasal Formulation
Phase I / II / III In vivo deposition FAST2CLINIC
Review Regulatory submission
Your bespoke R&D partner
in inhalation.
Flexible support across research, testing and analytical services from first formulation concept to clinical proof.
Discuss Your Project →News & Insights
Updates on inhalation science, regulatory developments, and i2c news.