Overview Of i2C Pharmaceutical Services

Gamma Scintigraphy
A unique offering within a CRO providing medical imaging for inhalation, nasal and oral delivery systems. Optional concurrent PK analysis can be conducted.
Laboratory Services
Provides a combination of formulation development, analytical testing, stability and small-scale process development for inhalation products.

Patented MDI Technology – Respitab™
An IP protected formulation technology for MDIs utilizing a unique propellant dispersible tablet providing robust formulations and performance.

Fast to Clinic
Small scale formulation manufacturing within a CRO allows for rapid manufacture, testing and release for next day dosing in Phase 1 studies.
Overview Of Formulation And Analytical Services
All work is performed in our state-of-the-art Cardiff facilities, ensuring responsive, client-centric service and robust support for clinical development.
Analytical & Manufacturing Capabilities
i2C stands out for its ability to deliver both laboratory and pilot-scale batches, supporting every stage of product development:

Small-scale GMP manufacturing of ‘micro’ batches for clinical studies, including gamma scintigraphy trials

Advanced pMDI production using Pamasol filling equipment, pressure mixing vessels, and vacuum crimpers

Powder blending for DPI manufacture (up to 2 kg) with Turbula blending technology

Comprehensive release testing with inertial impactors (NGI, Andersen, MSLI, MOUDI) and DUSA systems

Breathing simulation for nebuliser and spacer testing
Our flexible, scalable approach ensures efficient transitions from lab to clinic, with a proven track record in supporting innovative device and formulation projects.
Why Choose I2C?
Partner with i2C Pharmaceutical Services and experience the difference that expertise, innovation, and dedication can make for your inhalation product development.
Analytical Excellence
At i2C Pharmaceutical Services, analytical science is at the heart of everything we do. Our team provides the rigorous data and insight you need to accelerate your inhalation product development.
Comprehensive Analytical Services

Aerosol Characterisation:
Using industry-leading cascade impactors (NGI, Andersen, MSLI, MOUDI), we deliver detailed aerodynamic particle size distribution profiles for pMDIs, DPIs, and nebulisers, ensuring your product meets the highest standards for lung delivery.

Dose Uniformity & Delivered Dose Testing:
Our robust testing protocols guarantee consistent and accurate dosing from every inhaler, every time.

Potency & Content Uniformity:
We offer HPLC and spectroscopic assays to confirm active ingredient content and uniformity, supporting development and investigations.

Impurity Profiling & Stability Studies:
From forced degradation to Istability testing, our analytical scientists identify and quantify impurities.

Analytical Method Development:
We develop and optimise analytical methods tailored to your product requirements, ensuring smooth progression from early development to proof of concept.
Why i2C?
Choose i2C Pharmaceutical Services for analytical excellence that drives your inhalation innovation forward.
Inhalation And Nasal Product Development
From ab initio development to proof of concept, i2C supports every aspect of inhaled product development—including pMDIs, DPIs, nebulisers, and nasal sprays—tailoring each formulation to your molecule, device, and target patient profile.
Our Capabilities

Integrated Device & Formulation Expertise
Our team brings deep device knowledge, ensuring your formulation and inhaler work together for optimal drug delivery and patient usability.

Innovative Technologies
i2C offers proprietary platforms like Respitab®, a propellant-dispersible tablet technology that streamlines the formulation and scale-up of next-generation pMDIs—making development faster, simpler, and more cost-effective.

Flexible, Client-Focused Approach
Whether you need early proof-of-concept support, reformulation, or scale-up for clinical trials, i2C’s agile team delivers tailored solutions that reduce time to market and development risk.

Comprehensive Dosage Form Support
Our expertise spans powders, solutions, and suspensions for inhaled delivery, ensuring the right formulation for your product’s unique requirements.

Analytical Method Development:
We develop and optimise analytical methods tailored to your product requirements, ensuring smooth progression from early development to proof of concept.

Clinical & In Vivo Support
Our integrated services include bespoke gamma scintigraphy imaging studies to evaluate in vivo deposition, residence, and clearance—providing robust data for regulatory submissions and clinical success.
Why Partner with i2C?
Overview Of Gamma Scintigraphy
What is Gamma Scintigraphy?
Gamma scintigraphy is a non-invasive imaging technique that uses radiolabelled drug formulations to visualize and quantitatively assess how much of an inhaled medicine is deposited in the respiratory tract (or other target sites) after administration. It enables direct measurement of lung, oropharyngeal, and gastrointestinal deposition, as well as clearance and dispersion over time.
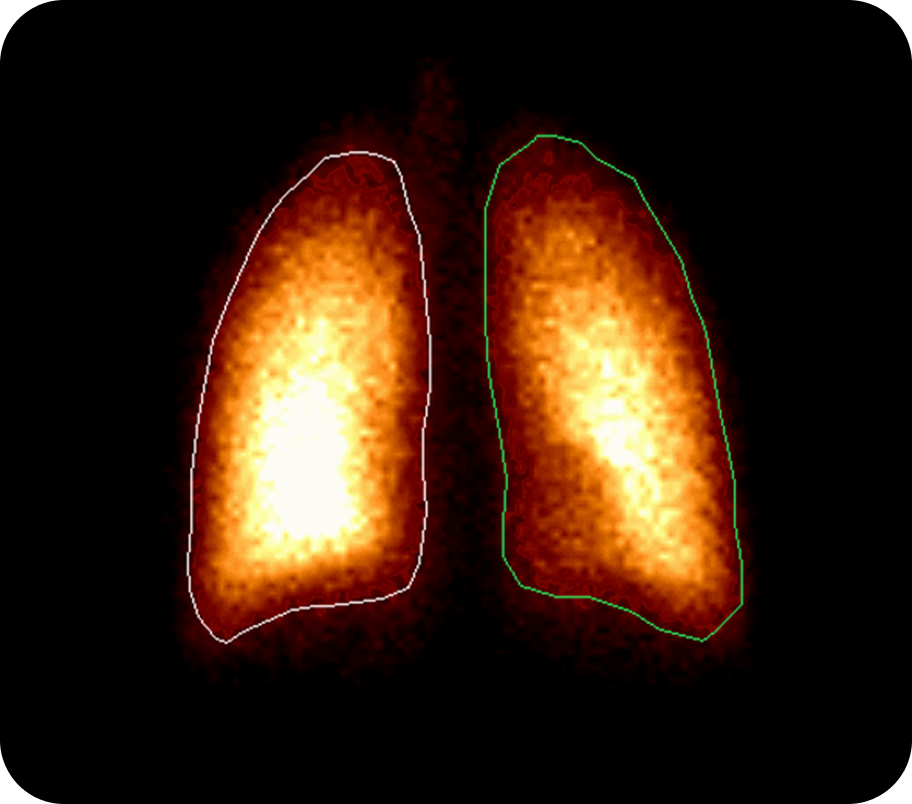
Key Service Highlights

Bespoke Clinical Studies
i2C designs and conducts tailored gamma scintigraphy studies for a wide range of inhalation devices—including pMDIs, DPIs, nebulisers, and nasal sprays—in both healthy volunteers and patient populations.

Quantitative Deposition Data
Provides direct, quantitative imaging of drug deposition in the lungs, oropharynx, and GI tract, supporting robust product characterisation and regulatory submissions.

Radiolabelling Expertise
i2C ensures radiolabelling methods accurately reflect the behaviour of the active drug, maintaining aerosol characteristics equivalent to commercial products.

Integrated Development Support
Gamma scintigraphy data is used to validate computational fluid dynamics (CFD) models, optimise formulation and device design, and support clinical and regulatory strategies.

Comprehensive Reporting
Full design, conduct, analysis, and reporting of studies—supporting both early-stage innovation and late-stage clinical evaluation.
Why Choose i2C?
In Summary :
i2C’s gamma scintigraphy services deliver the gold standard for in vivo evaluation of inhaled medicines—providing the quantitative data and regulatory confidence needed to accelerate your inhalation product from concept to clinic.
